|
12/23/2023 0 Comments Dichloromethane non reactivity 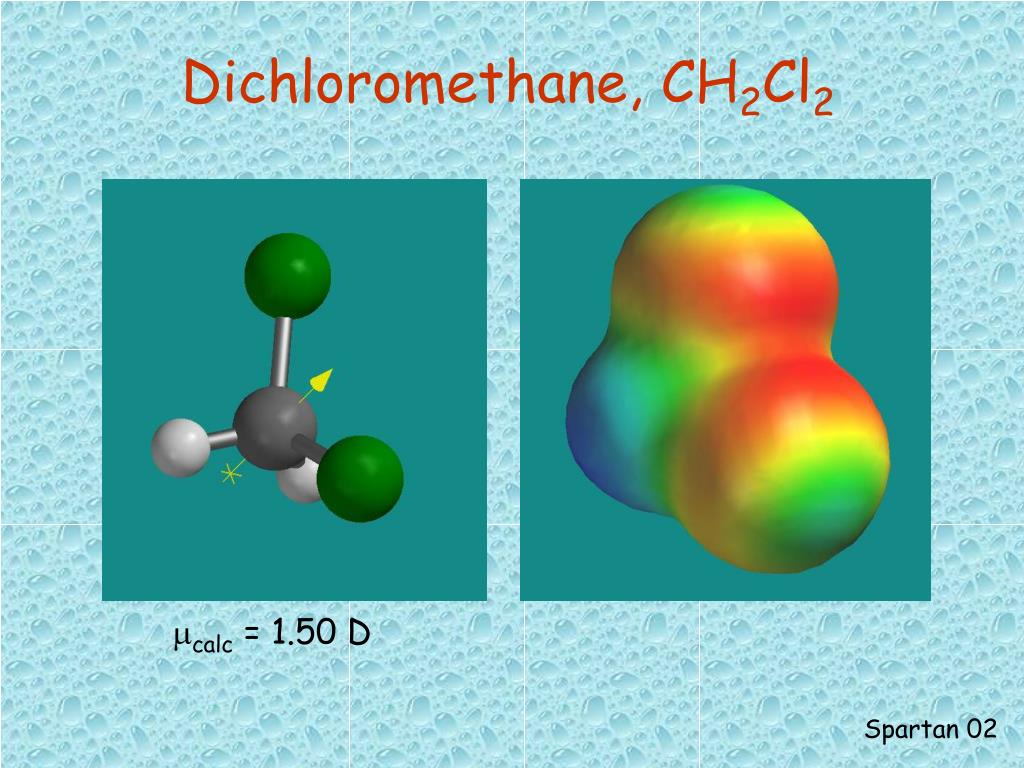
In general a carbene is neutral and has 6 valence electrons, 2 of which are non bonding.\) for Mechanism \(B\):Ĭustomarily, \(\nu\) is expressed in moles of product formed per liter of solution per unit of time (most frequently in seconds). These persistent carbenes are stabilized by a variety of methods often including aromatic rings or transition metals. However, there do exist so called persistent carbenes. The reactions of this section only deal with these short lived carbenes which are mostly prepared in situ, in conjunction with the main reaction. While the CHCl-amine reactions seem to proceed through nucleophilic attack at the methylene carbon atom, the reaction between dichloromethane and Bu3P is not. Other species that will also react with alkenes to form cyclopropanes but do not follow the formula of carbenes are referred to as carbenoids.Ĭarbenes were once only thought of as short lived intermediates. Methylene, H 2C, is simplest carbene, and in general carbenes have the formula R 2C. One common method of cyclopropane synthesis is the reaction of carbenes with the double bond in alkenes or cycloalkenes. This material is not reactive under normal ambient conditions. This structural difference affects their reactivity and chemical behavior. The use of dichloromethane and 1,2-dichloroethane resulted in a drop in enantioselectivity, whereas highly polar solvents, such as alcohols, resulted in a lack. Chloroform has three chlorine atoms attached to a central carbon atom, while dichloromethane has two chlorine atoms. Additionally cyclopropanes are present in numerous biological compounds. Both chloroform and dichloromethane are halogenated hydrocarbons, meaning they contain halogen atoms (chlorine) bonded to carbon atoms. The highly strained nature of cyclopropane compounds makes them very reactive and interesting synthetic targets. As shown in Table 1, ditritylation product 4 is favored over mono-tritylated product 3a when the non-polar dichloromethane was used as solvent (entry 1). Note that the deprotonation of chloroform generates the trichloromethanide anion, which spontaneously expels the chloride anion. The reaction was also tested on four nucleoside derivatives that have primary alcohols. The detailed mechanism of the formation of dichlorocarbene is given below. (14) Typically DCM is chosen for its versatility as a solvent, while the pyridine acts as a nucleophilic catalyst or a proton acceptor. dichloromethane as solvent and tetrabutylammonium tetrafluoroborate (TBABF4) or tetrabutylammonium hexafluorophosphate (TBAPF6) as supporting electrolyte. In particular, dichloromethane and chloroform (trichloromethane) play a key role as they find a plethora of applications (DCM) in industrial chemistry. 1998), 38 of Malays, 16 in India (Lee et al. Dichloromethane (DCM) and pyridine derivatives are frequently used together in reactions such as alcohol protection and acylation, (1-10) DessMartin oxidation, (11-13) and ozonolysis. PDF Tools Share Summary Chlorine derivatives of methane belong to the most important commodity chemicals among the halogenated hydrocarbons. Chlorine, which is similar to fluorine but not as reactive, was prepared by. Lithium reactivity with electrolytes leads to their continuous consumption and dendrite growth, which constitute major obstacles to harnessing the tremendous energy of lithium-metal anode in a. Dichloromethane (DCM) or methylene chloride is a volatile, colorless liquid with a chloroform-like odor. Due to its high reactivity, it is commonly found in nature bonded to many different elements. It is very reactive and is widely used for many purposes, such as as a disinfectant. 1994), 60 of the Chinese and Korean populations (Nelson et al. Chlorine is a halogen in group 17 and period 3. 
\)Ī carbenoid is best considered to be a reagent which, while not actually a carbene, behaves as if it were an intermediate of this type.ĭichlorocarbenes can also form cyclopropane structures and are created in situ from reagents such as chloroform and KOH. For example, non-conjugators make up about 11 of the Swedish population (Warholm et al. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
